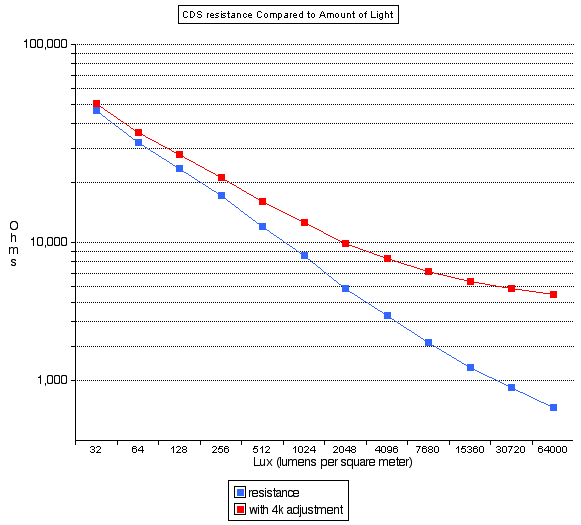
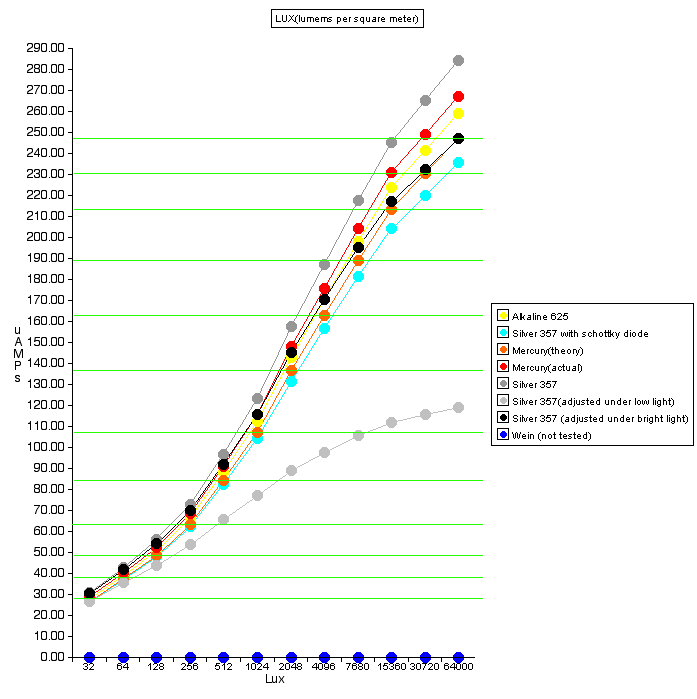
"The Minolta SR-T 101 uses a 1.35v, button-shape mercury battery for photo-graphic applications (Mallory PX-625, PX-13, Eveready EXP-625, EPX-13 or equivalent). "These mercury batteries your SRT was designed to use are now very hard to locate, as production has been discontinued in most of the world for very good environmental reasons. There are a number of possible replacements, but none of them result in exactly the same meter readings as the original design, at least without some effort at recalibration. This is because the meter is a very simple circuit which depends upon a specific stable voltage source.
| possibility | availability |
voltage characteristics |
relative lifetime voltage variation |
voltage changes with current
variation |
resistance to voltage variation
with temperature |
plus |
minus |
| Schottky diode adapter with
357 type silver oxide button cell |
357 batteries are available
almost anywhere. You can build your own adapter or purchase
the closely related MR9 (which I have not tested). |
Briefly 1.4 Nominally 1.35 Quick drop off at end of life |
more stable than mercury |
some |
good |
May be cheaper than 76
type photo batteries, may not require recalibration. |
Expensive, unless you make your
own adapters, which is not difficult to do. |
| 357 cell with O-ring to fit |
357 batteries are available
almost anywhere |
Briefly 1.6 Nominally 1.55 Quick drop off at end of life |
more stable than mercury |
no |
good |
Simple, easy and cheap.
May be cheaper than 76 type photo batteries. |
I've had some problems with
o-rings interfering with good electrical contact. Requires
recalibration since the voltage is so far off. |
| imported mercury replacements |
hard to come by, apparently
illegal to sell in many places |
1.4 or 1.35 volts, the energizer
technical page specifies both. The import I recently tested
read 1.46 volts under load! |
very stable |
no |
poor low temperature performance
in some situations |
original technology |
Apparently illegal to sell,
mercury is very toxic, it is unclear whether voltage is 1.35
or 1.4 volts. Perhaps both versions exist. |
| Schottky diode adapter with
76 type silver oxide button cell |
76 batteries are available almost
anywhere. You can build your own adapter or purchase the closely
related MR9 (which I have not tested). |
Briefly 1.4 Nominally 1.35 Slow decline towards end of life |
Possibly less stable than 357
variation and mercury. I've read somewhere that the charts on energizer.com
are no longer accurate and they now have stable voltages over their
lifetime. |
some |
good |
Expensive, unless you make your
own adapters, which is not difficult to do. |
|
| 76 type cell with O-ring to
fit |
76 batteries are available almost
anywhere |
Briefly 1.6 Nominally 1.55 Slow decline towards end of life |
Possibly less stable than 357
variation and mercury. I read somewhere that the charts on energizer.com
are no longer accurate and they now have stable voltages over their
lifetime. |
no |
good |
simple, easy and cheap |
I've had some problems with
o-rings interfering with good electrical contact. Requires
recalibration since the voltage is so far off. |
| zinc-air cell example Energizer (AC675)
or Wien MRB625 |
available in photo specialty stores
only |
Briefly 1.35 Nominal 1.3 Quick drop off at end of life |
Voltage varies depending upon recent
usage for 675 cells. I haven't tested Wien cells. |
Voltage varies depending upon recent
usage for 675 cells. I haven't tested Wien cells. |
good, but at high temperature the
lifetime is shorter |
Batteries are only good for a few
months. I haven't tested Wien cells. |
|
| alkaline 625 | available almost anywhere |
1.5 Slowly declines over lifetime of battery |
voltage varies greatly over
life of this battery - basically unusable for this application |
no |
good |
same form factor as original |
Variation in voltage over life
of battery makes this a less desirable solution than others |
| modify camera electronics with
voltage regulator |
how good at electronics design
are you? |
whatever you choose |
most stable |
should not |
depends upon circuit |
Requires camera modifications.
lowers lifetime of selected battery quite a bit, do to increased
current draw |

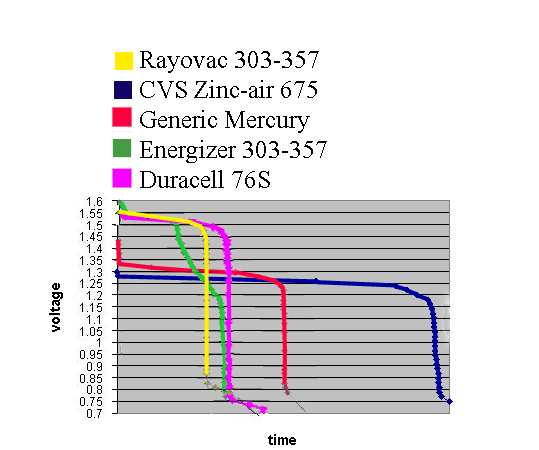
| CVS Zinc-Air |
Eveready 303-357 |
Rayovac 303-357 |
Generic Mercury |
Duracell 76 S |
|
| Measured milliamp hours | 486 | 145 |
136 |
252 |
173 |
| Expected lifetime in days with continous 200
uAmp load |
96 days*** |
30 days |
28 days |
53 days |
36 days |
| Brand new battery measured no load voltage | 1.388 | 1.62 |
1.598 |
1.447 |
1.591 |
| Brand new battery measured voltage with 2 milliamp load | 1.25 volts | 1.60 |
1.561 |
1.427 |
1.569 |
| Measured voltage drop over useful lifetime* | .059 volts | .27 volts |
.059 volts |
.1 volts |
.098 volts |
| Calculated internal resistance | 50 ohms | 9 ohms |
18.5 ohms |
10 ohms |
11 ohms |
| Calculated average voltage with 50 uAmp load | 1.35 | 1.35 |
1.56 |
1.30 |
1.53 |
| Calculated average voltage with 200 uAmp load | 1.34 | 1.35 |
1.55 |
1.30 |
1.52 |
| Calculated average voltage with 400 uAmp load | 1.33 | 1.35 |
1.55 |
1.3 |
1.52 |
| Calculated broken in battery voltage with 200 uAmp load** | 1.37 | 1.49 |
1.59 |
1.34 |
1.54 |
| Calculated old battery voltage with 200 uAmp load** | 1.31 | 1.21 |
1.53 |
1.24 |
1.48 |